Location: Home >> Detail
TOTAL VIEWS
Adv Geriatr Med Res. 2019;1:e190012. https://doi.org/10.20900/agmr20190012
1 Faculty of Physical Activity Sciences, Department of Kinanthropology, University of Sherbrooke, Sherbrooke, QC, J1K 2R1, Canada
2 Research Center on Aging, CIUSSS de l’Estrie-CHUS, Sherbrooke, QC, J1H 4C4, Canada
3 Research Center of the Centre Hospitalier Universitaire de Sherbrooke and Department of Medicine, Division of Endocrinology, University of Sherbrooke, Sherbrooke, QC, J1H 5N4, Canada
* Correspondence: Eléonor Riesco, Tel.: +1-819-780-2220 (ext. 45240) or +1-819-821-8000 (ext. 63337).
Background: It was reported that cow’s milk-based supplementation after resistance training reduced fat mass in older men. The reasons behind this beneficial impact remain to be elucidated. Hence, the aim of this study was to determine the effect of a cow’s milk-based supplementation after resistance exercise on lipid oxidation and systemic fat mobilization in older men.
Methods: Older men (age range: 60–75 years) participated in a randomized, double-blind, crossover study with the two following conditions: (1) Resistance exercise + Cow’s milk (C-milk) and (2) Resistance exercise + Rice milk (R-milk; isocaloric control). During a 180-min post-exercise period, energy expenditure, substrate oxidation (indirect calorimetry), plasma levels of glycerol (systemic fat mobilization) and free fatty acids (FFA) were measured.
Results: During the 180-min post-exercise period, lipid oxidation remained similar in both conditions. Glycerol levels decreased similarly in C-milk and R-milk (p ≤ 0.045). Although FFA levels increased progressively from 60 min to 180 min post-exercise in both conditions, the magnitude of changes (∆) was greater in C-milk condition between 60 and 120 min post-exercise (p < 0.018).
Conclusions: These results do not support a large impact of cow’s milk supplementation after resistance exercise on lipid oxidation and systemic fat mobilization. However, the greater increase in FFA levels suggests that re-esterification may be influenced.
C-milk, cow’s milk; EAA, essential amino acids; FFA, free fatty acids; I.Cal, indirect calorimetry; R-milk, rice milk; RMR, resting metabolic rate
Aging is associated with a progressive decline in muscle strength and mass as well as changes in muscle quality caused by a net reduction in protein synthesis [1]. In addition, reduction in muscle quality typically occur with an increase in fat mass [2]. These alterations play a significant role in the development of metabolic problems and progressive functional decline, increasing the risk of morbidity and mortality with aging [3,4]. Several factors can explain these modulations in body composition, among them, the reduction of physical activity level [5], the rise of sedentary behaviors and the deficiency in dietary protein [6]. Likewise, a decrease in resting energy expenditure can be seen [7], leading to an increased intramuscular lipids and a decreased lipid hydrolysis in adipose tissue [8]. Resistance exercise is generally considered to be a reliable strategy to promote muscle mass gain [9] and increase muscle strength [10]. Moreover, long-term resistance exercise training has been demonstrated to upregulate adipose tissue lipolysis and enhance energy expenditure in men [11].
Several mechanisms explaining the effects of protein consumption on fat metabolism have already been extensively explored, such as satiety hormones and food thermogenesis [12]. We recently showed that resistance training combined with a post-exercise protein supplement from cow’s milk (13.5 g of protein; 7 g of essential amino acids; EAA) reduced fat mass in older men [13]. These results are in line with those of a meta-analysis, which revealed a greater fat mass loss and lean mass increase when combining protein supplementation and resistance training [14]. Although the mechanisms explaining the effects of combining protein supplementation and resistance exercise on fat mass remained unclear, it is interesting to note that lipid oxidation and fat mobilization (adipose tissue lipolysis) are both acutely stimulated after one single bout of resistance exercise [15,16]. Moreover, it was demonstrated that leucine, an EAA found in cow’s milk, promotes a decrease of energy storage in adipocytes, an increase of lipid oxidation in muscle [17] and fat mass loss [18]. Therefore, we hypothesized that a greater fat mobilization and lipid oxidation would occur when cow’s milk supplementation is consumed following a single bout of resistance exercise. The aim of this pilot study was to test this hypothesis.
Because this is an exploratory pilot study, and to remove inter-individual variability, a randomized, double-blind, crossover (within-subject) study was performed with 8 men aged between 60 and 75 years (62.1 ± 1.4 years). To be included, participants had to meet the following criteria: (1) non-smoker, (2) physically active (≥75 min of vigorous intensity activity/week or ≥150 min moderate intensity activity/week), (3) null or moderate alcohol consumption (≤2 drinks/day), (4) apparently healthy and without medical treatment that can influence metabolism.
Study ProtocolAfter a phone screening, participants who met the criteria were invited for a baseline visit at the Research Centre on Aging. Resting metabolic rate (RMR) and substrate oxidation (indirect calorimetry), anthropometry (body weight, height, waist circumference), body composition (DXA), lipid profile, glucose and insulin levels were measured after a 12 h-overnight fast and 48 h without exercise. After a standard breakfast, physical activity levels (PASE questionnaire) were estimated and maximal strength (1-repetition maximum on leg press and lateral pulldown; 1-RM) was measured. At the end of this visit, specific instructions to record daily dietary intake for 3 non-consecutive days were given.
Among the eighteen men contacted by phone, only ten met the inclusion criteria and two were excluded after the first visit based on fasting glucose levels and high insulin levels (prediabetes). Hence, eight men completed this randomized double-blind crossover study. After the baseline visit, each participant was invited to two visits scheduled over two weeks to randomly perform two different experimental conditions: (1) Resistance exercise + Cow’s milk-based supplement (C-milk); (2) Resistance exercise + Rice milk-based supplement (R-milk; isocaloric without EAA). Each post-exercise beverage (C-milk or R-milk) was consumed within 5–10 min after the end of the exercise session in a double-blinded fashion (participant and experimenters).
Prior to each condition, participants were asked to avoid exercise or strenuous physical activity for 48 h. For each condition, all measures were performed in the same manner as depicted in the Figure 1. During the 180 min post-exercise period, energy expenditure and lipid oxidation (determined by indirect calorimetry; I.Cal) were measured and blood samples were collected to estimate systemic fat mobilization (plasma glycerol [19,20]) and measure free fatty acids (FFA), glucose and insulin levels. Three measures of energy expenditure and lipid oxidation were performed: (1) between 30–60 min after resistance exercise (I.Cal30–60); (2) between 90–120 min (I.Cal90–120); (3) between 150–180 min (I.Cal150–180). Five blood samples were collected to measure glycerol, FFA, glucose, and insulin plasma levels: (1) Before resistance exercise (T−60); (2) immediately after the end of resistance exercise + post-exercise supplement (T0); (3) 60 min post-exercise (T60); (4) 120 min post-exercise (T120) and (5) 180 min post-exercise (T180).
The study was conducted according to the guidelines laid down in the three principles of Tri-Council policy statement and all procedures were approved by the Ethics Committee of the Centre Intégré Universitaire de Santé et de Services Sociaux de l’Estrie (CIUSSS de l’Estrie-CHUS). Written informed consent was obtained from all participants before entering the study.
Resistance Exercise SessionThe exercise session was the same for both experimental conditions and was held at our laboratory under the supervision of an exercise specialist. It consisted of a 5-min warm-up and a 5-min cool-down on a treadmill at low intensity and ∼45 min of resistance exercise using dynamic exercises performed with weighted pulley machines (Life fitness, Canada), free weights or body weight. The program included 12 exercises organized in 3 circuits of 4 exercises (for upper and lower body) with a 4-min break between each circuit. Participants had to perform 3 sets of 8 repetitions at 75%–80% 1-RM (based on leg press and lateral pulldown maximal strength test) for loaded exercises and 3 sets of 15 repetitions for bodyweight exercises.
Standardization of Food Intake before Each Experimental ConditionTo avoid any impact of food intake on substrate oxidation and metabolic measures, each participant received a lunch box designed by a nutritionist. This lunch box consisted of a lunch and a dinner for the day before the visit, as well as a breakfast for the morning of the experimental visit (7:00 am). All participants were instructed to consume all but only the food included in the lunch box. As an example, a person with an energy requirement close to 2300 kcal/day received a lunch box (2288 kcal) with 288.4 g of carbohydrates, 93.4 g of lipids and 88.3 g of protein to meet the Dietary Reference Intakes (Health Canada).
Energy requirements were estimated for each participant using the following equation: Energy requirements (kcal/day) = RMR (kcal/day) × 1.6 (physical activity level factor for a moderately active individual [21]).
Post-Exercise SupplementationAll participants were asked to consume the supplement (C-milk or R-milk) 5–10 min after the end of the exercise session. Both supplements, each of 375 mL, were isocaloric chocolate-flavored beverages. All drinks were prepared by an independent investigator and served in opaque cups with a lid with respect to the double-blind design. Supplements made from cow’s milk (7 g EAA) and rice milk (0 g EAA) were prepared using the same recipes as previously used in our laboratory [22].
Resting Metabolic Rate, Energy Expenditure and Respiratory Exchange Ratio (RER)Resting metabolic rate (RMR; kcal/day) as well as RER, used for estimating relative lipid and carbohydrate oxidation, were measured after a 12-h overnight fast by indirect calorimetry using a respiratory mask (CCM Express system, Medgraphics Cardiorespiratory Diagnostic, St Paul, MN, USA). During the 180-min post-exercise recovery, energy expenditure (kcal/day) and RER were also measured at three different time points ((1) I.Cal30–60, (2) I.Cal90–120, (3) I.Cal150–180) using the same procedures. Measurements lasted 30 min with the participant lying comfortably on their back. Excluding the first 10 min and the last 5 min, RMR and energy expenditure were then calculated with the remaining 15 min using the Weir equation [23]. Relative lipid and carbohydrate oxidation (%) was calculated with the RER using the Péronnet & Massicotte non-protein table with the assumption that protein oxidation is negligible [24]. It was previously demonstrated that post-exercise amino acid oxidation (leucine) increases only when protein ingestion exceeds 20 g [25], which is greater than what was contained in both supplements used in the present study (Table 1). Absolute lipid and carbohydrate oxidation (g/min) were calculated using stoichiometric equations [24]. In our laboratory, test-retest measures of RMR in 10 adults, with a 1-week interval, yielded a mean absolute coefficient of variation of 2.1%.
Metabolic Measures during Post-Resistance Exercise RecoveryBefore each experimental condition and during the 180 min resting period, blood samples were collected by an experienced research nurse in EDTA-containing Vacutainer tubes to measure plasma glycerol, FFA, glucose and insulin levels. For the 180 min recovery period only (T0, T60, T120, and T180), a catheter was inserted (at T0) in the antecubital vein to allow the collection of blood samples.
Plasma levels of glycerol were measured with a colorimetric method (EnzyChrom™ Glycerol Assay Kit, BioAssay Systems, Hayward, CA, USA) and used as a proxy measure of systemic fat mobilization because it was previously demonstrated that plasmatic glycerol levels are a good index of systemic lipolysis [26] as it is strongly related to its rate of appearance [19,20]. Since FFA availability influences lipid oxidation in muscle [27,28], we also measured plasma FFA levels by an enzymatic method (ESBE Scientific, Markham, ON, Canada). Glucose metabolism was assessed by plasma glucose (autoanalyzer Yellow Spring Instrument, Yellow Springs, OH, USA) and insulin (enzymatic and immunological method, CHUS laboratory, Sherbrooke, QC, Canada) levels.
Anthropometry and Body CompositionParticipant’s height was measured using a wall stadiometer (Takei, Tokyo, Japan), body weight with an electronic scale (SECA707, Hamburg, Germany). Waist circumference was measured at the upper edge of the hip bone using a measuring tape. Body composition was measured using the dual-energy X-ray absorptiometry method (DXA; GE Prodigy Lunar, Madison, WI, USA), allowing the measurement of bone mass, fat mass (FM) and lean body mass (LBM) in upper and lower limbs and trunk. Coefficients of variation in our laboratory, for repeated measures (1 week apart) of FM and LBM in 10 adults are 0.9% and 0.5%, respectively.
Physical Activity LevelPhysical activity level was assessed using the Physical Activity Scale for the Elderly (PASE) questionnaire [29]. Participants reported leisure time, household and work-related activities during the past week. Daily activities were first scored according to the intensity and duration of reported activities and then summed to produce a global score representing physical activity level (range of scores between 0–793), where higher score corresponds to a high level of physical activity [30].
Statistical AnalysesThe normality of variables was verified using the Shapiro-Wilk test and normal probability plots. Due to abnormality in the distribution of several variables and the small sample size, non-parametric statistics were used, following statistical considerations [31]. Data are presented using the median and interquartile range in figures as well as in tables. Friedman tests were performed in each condition to measure changes in each variable. All p values for Friedman tests were corrected with the False discovery rate procedure, using the Benjamini-Hochberg procedure. Comparison of each condition at each time point was performed with Wilcoxon tests. All analyses were performed using SPSS 21.0 program for windows (SPSS, Chicago, IL). Statistical significance was set at p ≤ 0.05.
Baseline characteristics are shown in Table 2.
Energy expenditure progressively decreased in C-milk condition and tended to decrease in R-milk during the 180 min recovery (R-milk; p = 0.06 and C-milk; p = 0.01). In R-milk, energy expenditure seemed to be maintained until I.Cal90–120 and decreased significantly between I.Cal90-120 and I.Cal120–180 (p = 0.03). In C-milk, energy expenditure decreased significantly from I.Cal30–60 to I.Cal120–180 (p = 0.05).
As shown in Figure 2, lipid oxidation demonstrated no significant changes in the first hour after resistance exercise. However, from I.Cal90–120 to I.Cal120–180, lipid oxidation increased significantly in both conditions (R-milk: p = 0.03 and C-milk: p = 0.05).
Systemic Fat Mobilization (Glycerol Levels) and FFA LevelsGlycerol levels increase significantly for both conditions from baseline (T−60) to post-exercise (T0)(all p < 0.05). During the first 120 min recovery (T0 to T120), the levels decreased similarly in both conditions (p < 0.045), with no significant difference between conditions (Figure 3A). Then, between T120 to T180, glycerol levels increased in R-milk only (p = 0.046).
Plasma FFA levels increased significantly (p < 0.04) and similarly in response to resistance exercise (from T−60 to T0) in both conditions. During the first hour of recovery (from T0 to T60), FFA levels dropped significantly in both R-milk (p = 0.01) and C-milk (p = 0.017). From T60 to T180, FFA levels increased significantly in both conditions. Between T60 and T120 (p = 0.01) and also between T120 and T180 (p = 0.01), FFA levels increased in C-milk while in R-milk, FFA levels increased significantly only between T120 and T180 (p = 0.03; Figure 3B). Furthermore, changes (∆ values) in C-Milk between T60 and T120 was significantly different from R-milk (p = 0.02).
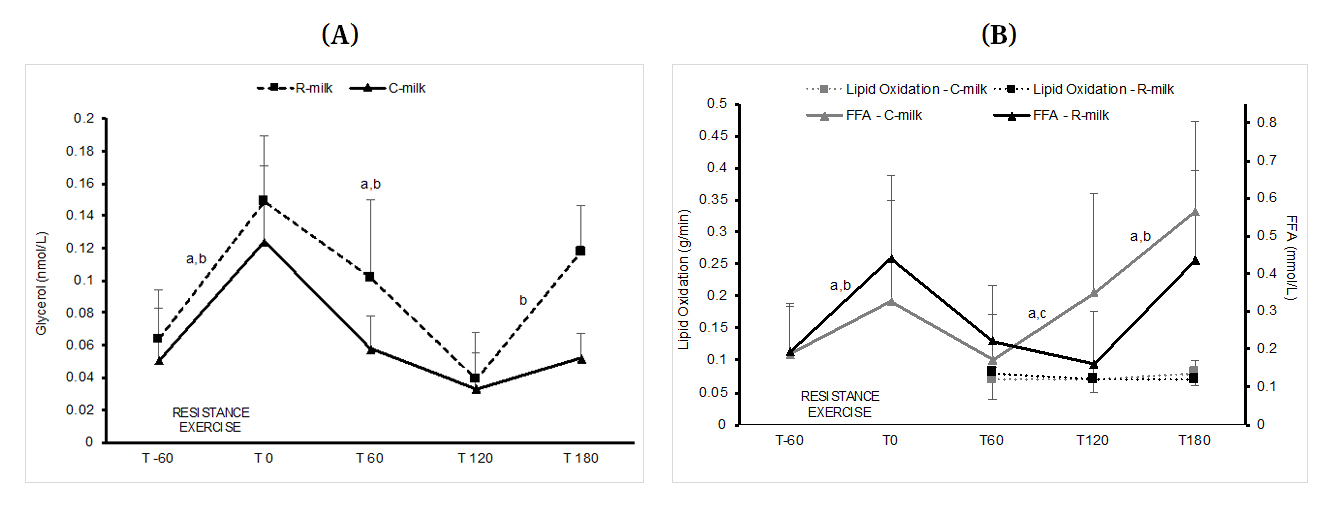 Figure 3. (A) Changes in plasma glycerol levels during the post-exercise recovery. Data are presented with median and interquartile range; a: significant changes in C-milk condition only (p < 0.05); b: significant changes in R-milk condition only (p < 0.05). (B) Lipid oxidation and plasma FFA levels during and after resistance exercise. Data are presented with median and interquartile range; a: significant changes in C-milk condition only (p < 0.05); b: significant changes in R-milk condition only (∆ values; p < 0.05); c: significant differences between condition (∆ values; p < 0.05).
Figure 3. (A) Changes in plasma glycerol levels during the post-exercise recovery. Data are presented with median and interquartile range; a: significant changes in C-milk condition only (p < 0.05); b: significant changes in R-milk condition only (p < 0.05). (B) Lipid oxidation and plasma FFA levels during and after resistance exercise. Data are presented with median and interquartile range; a: significant changes in C-milk condition only (p < 0.05); b: significant changes in R-milk condition only (∆ values; p < 0.05); c: significant differences between condition (∆ values; p < 0.05).
As shown in Table 3, glucose levels were similar between both conditions before and after resistance exercise (T0)(all p ≥ 0.3). From there, glucose levels in the R-milk condition remained stable while they decreased in C-milk to reach a significant difference between both groups at T60 (p = 0.01). At T120, glucose was lowered in R-milk, while still decreasing in C-milk compared to T60. From T120 to T180, glucose levels remained stable in both conditions without significant differences between conditions (p = 0.9).
Insulin levels increased significantly during the first hour (from T0 to T60; p = 0.02) and decreased between T60 to T120 (p = 0.02) in R-milk. A similar pattern was observed in C-milk condition without reaching a statistically significant threshold (from T0 to T60: p = 0.12). However, a significant decrease in insulin levels was observed from T60 to T180 in C-milk (from T60 to T120: p = 0.03 and from T120 to T180: p = 0.02).
The main objective of the present study was to examine the acute effect of a cow’s milk-based supplement consumed after resistance exercise on lipid oxidation and fat mobilization in older men. Our hypothesis was that cow’s milk-based supplement (containing 7 g of EAA) consumed after resistance exercise would increase fat mobilization and lipid oxidation during the recovery compared to an isocaloric non-protein supplement. Our results showed that the C-milk supplementation provided similar responses for fat mobilization and lipid oxidation when compared to an isocaloric non-protein supplement. However, the cow’s milk-based supplement induced an earlier increase in plasma FFA levels during post-exercise recovery.
Based on previous studies showing that both dairy calcium consumption [32] and resistance training [11] can upregulate adipose tissue lipolysis and enhance energy expenditure, we made the hypothesis that their respective effects (C-Milk condition) could be added to increase systemic lipolysis activity compared to the isocaloric non-protein supplement (R-milk condition). This would have also contributed to explain the previously observed fat mass loss in response to a 16-week intervention combining resistance training and cow’s milk-based supplementation in older men [13]. However, while systemic lipolysis activity was stimulated by resistance exercise, as shown by glycerol levels, it was relatively low during the recovery period, with no difference between both conditions.
After 30 min of recovery, lipid oxidation started to increase when post-exercise cow’s milk-based supplement was consumed. FFA levels also increased earlier during the recovery period in this condition (C-Milk) compared to the isocaloric non-protein supplement. However, FFA levels increased while glycerol levels remained low and stable. Interestingly, lipolysis seemed to be increased mainly in R-milk condition after 120 min of recovery, as glycerol levels abruptly rose compared to C-milk condition, while being consistent with FFA increase. Considering the strong inhibitory effect of insulin on lipolysis, this phenomenon may be the direct effect of the substantial decrease in this circulating hormone levels between 60 min and 120 min of recovery in the R-milk codition.
Although measuring glycerol levels as a proxy for lipolysis activity may not be as accurate as direct methods [33], these results nevertheless suggest that the increased FFA levels observed during the recovery period were not related to a higher lipolytic rate [11,34]. Hence, we hypothesized that FFA re-esterification by adipose tissue was reduced to respond to the metabolic demand. In line with this, it was demonstrated that leucine, an EAA found in cow’s milk, inhibits lipid storage in adipocytes [35]. Therefore, these results may suggest that resistance exercise combined with dairy supplementation might affect lipogenesis by reducing FFA re-esterification in order to favor lipid oxidation in muscle. This would be in agreement with a previous study showing that dairy calcium consumption reduces calcitriol (1,25-hydroxyvitamin D) levels, which decreases adipocyte intracellular calcium influx, and thus reduce lipogenesis [32]. On a chronic basis, this could contribute to a loss of fat mass. Nevertheless, it cannot be ruled out that the chronic effects of resistance exercise may also contribute to stimulating adipose tissue lipolysis [36]. Future studies using adipose tissue biopsies and measures of fatty acid re-esterification enzymes in adipose tissue (e.g., PEPCK) should be performed to confirm this hypothesis.
Some limits of this study deserve to be mentioned. Considering the exploratory aspect of this study and the fact that both experimental conditions were performed by each participant, using a proxy measure of fat mobilization was ethically more suitable than using more invasive techniques such as adipose tissue biopsies or a combination of isotopic ([d5] glycerol) and microdialysis techniques. Moreover, for each condition and each participant, indirect calorimetry was performed in fasting state at rest and not right before each condition, which means that data for lipid oxidation is unavailable prior to resistance exercise and supplementation. This limitation makes it difficult to interpret the results obtained for lipid oxidation compared with baseline.
Still, some strengths of the study are worth discussing. The advantage of such crossover design is that each participant acts as his own control and this significantly reduces inter-individual variability. Furthermore, the fact that all participants, as well as the experimenters and exercise physiologist, were blinded with regards to the supplement reduced potential bias. Finally, for all participants, energy intake was controlled at each condition, using standardized meals (for lunch, dinner, and breakfast) based on individual indirect calorimetry-based energy expenditure estimation.
To our knowledge, this is the first study to investigate the acute effect of post-exercise cow’s milk-based supplementation on lipid oxidation and fat mobilization in older adults. As aging-related reduced muscle strength and increased fat mass can ultimately lead to serious health conditions such as sarcopenia and obesity [37–39], combining resistance exercise and dairy supplementation may represent an efficient strategy for older adults.
In conclusion, our results suggest that cow’s milk based as well as isocaloric non-protein supplementation after resistance exercise do not strongly affect lipid oxidation in elderly men in the hours following exercise. However, it should not be ruled out that a reduced FFA re-esterification may contribute to a decrease in fat mass and explain the chronic impact of cow’s milk-based supplement combined with resistance exercise that was previously reported. Further research is needed to grasp underlying mechanisms of long-term resistance exercise and dairy supplementation-induced loss of fat mass.
The dataset of the study is available from the authors upon reasonable request.
ER and IJD designed the study. TA and MD collected the data. AF analyzed the data and wrote the paper with input from all authors. DT contributed to the interpretation of data for the work and critically review the manuscript.
The authors declare that they have no conflict of interest.
The study was supported by the Quebec Network for Research on Aging.
We want gratefully thanks all the participants for their participation and contribution in this study. Special thanks to Martine Fisch, the research nurse, for her professional work and all exercise physiologist (kinesiologists) who supervised the exercise training protocol.
1.
2.
3.
4.
5.
6.
7.
8.
9.
10.
11.
12.
13.
14.
15.
16.
17.
18.
19.
20.
21.
22.
23.
24.
25.
26.
27.
28.
29.
30.
31.
32.
33.
34.
35.
36.
37.
38.
39.
Fontvieille A, Tremblay D, Amamou T, Du Bois-dit-Bonclaude M, Dionne IJ, Riesco E. Acute effect of post-resistance exercise milk-based supplement on substrate oxidation and fat mobilization in older men: A pilot study. Adv Geriatr Med Res. 2019;1:e190012. https://doi.org/10.20900/agmr20190012

Copyright © 2020 Hapres Co., Ltd. Privacy Policy | Terms and Conditions