Location: Home >> Detail
TOTAL VIEWS
Crop Breed Genet Genom. 2019;1:e190017. https://doi.org/10.20900/cbgg20190017
1 Borlaug Institute for South Asia (BISA), Ludhiana, Punjab 141004, India
2 National Bureau of Plant Genetic Resources, Pusa Campus, New Delhi 110012, India
3 Institute of Agricultural Sciences, Banaras Hindu University, Varanasi 221005, India
4 Leibniz Institute of Plant Genetics and Crop Plant Research (IPK), Corrensstr. 3, 06466 Gatersleben, Germany
5 CIMMYT, G-2, B-Block, NASC Complex, DPS Marg, New Delhi 110012, India
* Correspondence: Arun Kumar Joshi.
This article belongs to the Virtual Special Issue "Genetic Gains in Plant Breeding"
Spot blotch (causative pathogen the fungus Bipolaris sorokiniana) is a damaging disease of wheat in warm and humid environments, which are prevalent in the Indian subcontinent. Genetic analyses have indicated that resistance is mediated by a number of independent genes, each contributing a small to intermediate size effect, meaning that combinations of three to five genes are required to ensure a high level of resistance. Near-immunity to spot blotch is not known. Hence, in order to seek further genetic gain on resistance, populations were developed from four simple crosses between the resistance donors “Yangmai#6” and either “Mon/Ald”, “Chirya#3”, “Tia#1” or “Ning#8201” with the aim of stacking resistance genes for resistance in order to generating lines showing near-immunity to the disease. The strategy was evaluated by genotyping the selections at 14 microsatellite loci linked to spot blotch resistance genes. The resistance locus most frequently retained by the selections maps to chromosome 6D; the second most frequently retained one maps to chromosome 2B. The use of a donor × donor crossing strategy was effective for developing lines with near-immunity to spot blotch disease.
Spot blotch (caused by Bipolaris sorokiniana, teleomorph Cochliobolus sativus) is a damaging disease of wheat plants raised in warm, humid environments [1–3]. Nearly 9 Mha of the crop grown in the Indian subcontinent is considered to be at risk of infection [4], and this area will likely grow if predictions of global warming prove accurate [5]. The extent of yield loss caused by the disease is highly dependent on local climatic conditions, but can reach as high as 42% [6]. Based on the observation of crops grown in India, Nepal and Bangladesh, Saari [7] has estimated an average yield loss of 20%, matching the estimated of 18% reported by [8]. An extensive breeding effort devoted to developing cultivars expressing significant resistance against this disease. Gupta et al. [3] has resulted in the release of a number of viable cultivars, but none of these are completely immune [9,10]. Potential donors of resistance have been identified both within the primary gene pool [2,11,12] and in a number of synthetic hexaploid materials [13,14].
While it may not be possible to achieve full immunity against a hemibiotrophic pathogen such as B. sorokiniana, achieving near-immunity may be feasible. In any case, full immunity may not be a desirable goal, given the risk of a catastrophic breakdown in resistance, as has been experienced in a number of crop/pathogen systems e.g., Southern corn blight and wheat stem and stripe rusts [15]. In contrast, near-immunity tends to be more durable because it is generally based on multigenic control and does not impose high selection pressure on the pathogen [16]. Mapping the components of spot blotch resistance has led to the identification of a number of molecular markers linked to resistance-associated genes/quantitative trait loci (QTL)[3,17]. For instance, the greater part of the resistance displayed by “Yangmai#6” is controlled by four genes/QTL [18], that of “Mon/Ald” by two or three [19], that of “Ning#8201” by more than one [11,20] and that of “Chirya#3” by at least five [21]. “Yangmai#6” and “Chirya#3” both harbor the QTL Qsb.bhu-2B, while “Yangmai#6” and “Ning#8201” both carry QSb.bhu-2B and QSb.bhu-5B [21]. The allelism between some of these loci has also been elucidated [2,19,20,22]. Singh et al. (2015) used conventional crossing and selected bulk method to develop spot blotch resistant lines [2].
A series of genetic analyses has established that resistance to spot blotch disease in wheat is under the control of a number of genes, each contributing a small to intermediate sized effect [10,21,23]. Thus accumulating several of these genes represents the most feasible way of breeding for near-immunity [16]. The parents of the crosses used here were selected on the basis that they each exhibit a level of resistance [1,18,19,21]. The present study sought to employ a combination of conventional and marker-assisted breeding to develop lines expressing near-immunity to spot blotch resistance.
Four simple crosses were effected in the main crop season 2008-09: “Yangmai#6” × “Mon/Ald”, “Yangmai#6” × “Chirya#3”, “Yangmai#6” × “Tia#1” and “Yangmai#6” × “Ning#8201”. All four crossing parents (their pedigrees are given in Table 1) show some resistance to spot blotch disease. The genetic basis of resistance in “Yangmai#6”, “Chirya#3” and “Ning#8201” has been uncovered [18,21]. The F1 hybrids were raised in an off-season nursery in year 2009, and a population of ~3000 F2 progeny was grown in the field (2009–2010) at Banaras Hindu University (Varanasi, Uttar Pradesh, India). Between 400 and 500 F2 selections per cross were made based on their disease phenotype. Seed from individual F2 plant was harvested to grow F3 families in the main season of 2010–2011. The materials were advanced to F5 (2012–2013) with continued selection for resistance using the selected bulk approach [24]. Agronomically superior F5 plants were identified by visual inspection, and were then genotyped using the set of microsatellite markers (SSRs) described below.
Field ExperimentationThree replicates of each line of F4 (2011–2012) and F5 (2012–2013) were planted as two rows of 3 m, each spaced 20 cm apart. Following the suggestion of [18], in order to encourage the build-up and spread of inoculum, a row of the spot blotch disease susceptible cultivar “Sonalika” was included after every 20 progeny rows, as well as in the alleys. The trial was planted in late December to ensure that the post-anthesis stage occurred at a time when the temperature conditions were most conducive to the development of the disease [25]. Parental cultivars along with the progenies of respective crosses were also planted to serve as positive control.
Following best commercial practice, the plots were provided with 120 kg/ha N, 60 kg/ha P2O5 and 40 kg/ha K2O. The nitrogen application was split into three, where half was given at sowing, a quarter after the first irrigation (21 days after sowing) and a quarter following the second irrigation (40 days after sowing). The phosphorus and potassium components were provided in full at the time of sowing.
Inoculation of PathogenThe plots were artificially inoculated with B. sorokiniana spores, as described by Chaurasia et al. [11]. Artificial epiphytotic conditions were created by spraying a pure culture of B. sorokiniana (NABM MAT1; NCBIJN128877, BHU, Varanasi, India), which is known to be highly aggressive [17]. The isolate was obtained from the department of Plant Pathology and Mycology, BHU and multiplied on sorghum grain. The inoculation was done during evening hours using hand held sprayer at tillering and flag leaf emergence stages. The field was irrigated immediately after inoculation to maintain sufficient moisture for disease build-up.
Since the time required by the five parental lines to reach anthesis and maturity differed by up to eight days, the timing of the measurements needed to generate an area under disease progress curve (AUDPC) was based on physiological age (as measured by the Zadoks growth stage; Zadoks et al. [26] rather than on the number of days post planting [27]. Thus, disease severity (%) of each F4 and F5 lines was recorded at GS63 (beginning of anthesis to half complete), GS69 (anthesis complete) and GS77 (late milking), following the suggestion of [18]. A score of 0 was assigned for complete immunity and of 100 for full susceptibility. An AUDPC, based on disease severity scores, was calculated from the expression [27]:
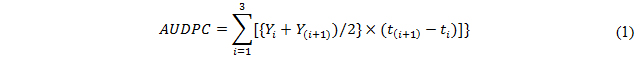
where Yi represented disease severity at time ti and t(i + 1) − ti the number of days which had elapsed between two consecutive observations.
GenotypingGenomic DNA, isolated from 15 day old seedlings following the protocol given by Kumar et al. [18], was used as template for a series of PCRs targeting a set of 14 SSR loci ([28] Table 2) known to flank genes/QTL determining spot blotch resistance [18,21]. The PCR performed in 0.1 mL tubes using 25 µL reaction mixture. Each reaction (25 µL) contained 25–100 ng template DNA, 250 nM of each primer (one of which was labeled with Cy-5), 200 µM dNTP, 1.5 mM MgCl2, 1× PCR buffer and 1 U Taq DNA polymerase. The reactions were subjected to an initial denaturation (92 °C/3 min), which was followed by 45 cycles of 92 °C/1 min, annealing temperature (50, 55 or 60 °C)/1 min, 72 °C/2 min, and were completed with a final extension step of 72 °C/10 min. The resulting amplicons were separated using an ALF express device (Amersham Biosciences Europe GmbH, Freiburg, Germany). Fragment sizes were extrapolated from the migration of four DNA fragments of known size (73, 122, 196 and 231 bp). Any pair of fragments estimated to differ in length by <2 bp was considered to represent two identical sequences (Supplementary Figure S1). The fragment sizes generated by each SSR assay from template of the various donors are listed in Table 2. Based on the known fragment size amplified by respective markers, the presence or absence of QTL was recorded.
An analysis of variance for AUDPC across the two consecutive seasons was performed using SAS statistical software (SAS Inc., Cary, NC, USA). The heritability (h2) of resistance was estimated from the expression [1 − [MS (genotype × year)]/MS (genotype), following Nyquist (1991) [29]. A Pearson correlation coefficient was calculated between the level of resistance shown by an F4 line and its F5 progeny using a routine implemented in SAS statistical software. For the purposes of the analysis of variance of resistance, genotypes were considered to have random effect. Paired t tests were conducted using the “T-Test Calculator” tool (www.socscistatistics.com/tests/studentttest/Default2.aspx).
The regression analysis of all individuals was performed to study the overall effect of allele accumulation on the level of disease resistance.
The AUDPC of the five donor lines ranged from 267.5 ± 18.6 (“Chirya#3”) to 331.7 ± 32.4 (“Yangmai#6”) in 2011–2012, and 191.4 ± 33.8 (“Mon/Ald”) to 365.0 ± 25.5 (“Tia#1”) in 2012–2013, while that of the susceptible check “Sonalika” was 922.5 ± 82.5 in 2011–2012 and 775.1 ± 30.0 in 2012–2013 (Table 1). A paired t test indicated that each of the donor lines behaved more or less consistently across the two seasons based on average disease severity (Table 1).
The selections made from three of the populations showed lower mean AUDPC in F5 except “Yangmai#6” × “Mon/Ald” (Figure 1, Table 3). “Yangmai#6” was generally associated with a higher AUDPC than the other three donors; the sole exception was “Tia#1” in the second year (Supplementary Figure S2a–h, Table 1). The “Yangmai#6” × “Ning#8201” F4 and F5 selections” mean AUDPCs were, respectively 153.3 ± 12.2 and 168.7 ± 18.0; the equivalent AUDPCs for the “Yangmai#6” × “Chirya#3” and “Yangmai#6” × “Mon/Ald” selections were 197.2 ± 4.7 and 187.6 ± 16.3, and 418.5 ± 17.9 and 375.0 ± 23.5, respectively. In each case, there was a significant contribution made by the environment (year) to the variance (Table 4). The h2 parameter ranged from 0.71 to 0.96 (Table 4). The intergenerational (F4 vs F5) correlations ranged from +0.57 (“Yangmai#6” × “Mon/Ald”) to +0.68 (“Yangmai#6” × “Chirya#3”) (Table 3).
The level of resistance shown by selections #14, #45 (harboring resistance genes/QTL on chromosomes 2B and 6D) and #75 (on chromosomes 2B, 6D and 7D) from the “Yangmai#6” × “Mon/Ald” population was lower than that of selections #1, #2, #10, #21, #42 and #58, each of which harbored either two or three genes/QTL (Supplementary Table S1). Meanwhile selections #16, #21 and #98 from the “Yangmai#6” × “Chirya#3” population harbored seven resistance genes and selections #1, #11, #63, #73 and #96 harbored six (Supplementary Table S2). The AUDPC values of selected lines in each cross was lower than the corresponding resistant parents. In the “Yangmai#6” × “Tia#1” population, all of the selections harbored one or more of the resistance genes/QTL carried by the parental lines (Supplementary Table S3).
Based on marker genotype, six loci can be expected to be segregating in the “Yangmai#6” × “Ning#8201” population (Supplementary Table S4) and seven in the “Yangmai#6” × “Chirya#3” (Supplementary Table S2). About one in six of the selections carried Qsb.bhu-5B. A regression analysis implied that the level of resistance to spot blotch depends on the number of distinct resistance genes present (Figure 2). For instance, the average AUPDC of the “Yangmai#6” × “Mon/Ald” selections harboring four or more genes/QTL was almost one-half that of the average of plants harboring two (214.58 vs 472.50, see Supplementary Table S1). The AUDPC of the three most resistant selections in each population was superior to that of the better parent: for instance, the AUDPC of “Yangmai#6” × “Mon/Ald” selection #58 was 18.4% lower than that of better parent “Mon/Ald”, while that of “Yangmai#6” × “Ning#8201” selection #1 was 57.7% lower than that of Ning#8201 (Table 5). To estimate the number of genes/alleles in finally selected lines (F4 and F5) the χ2 analysis was performed. The analysis confirmed that all the selected lines included higher number of genes/QTL determining resistance (Table 6).
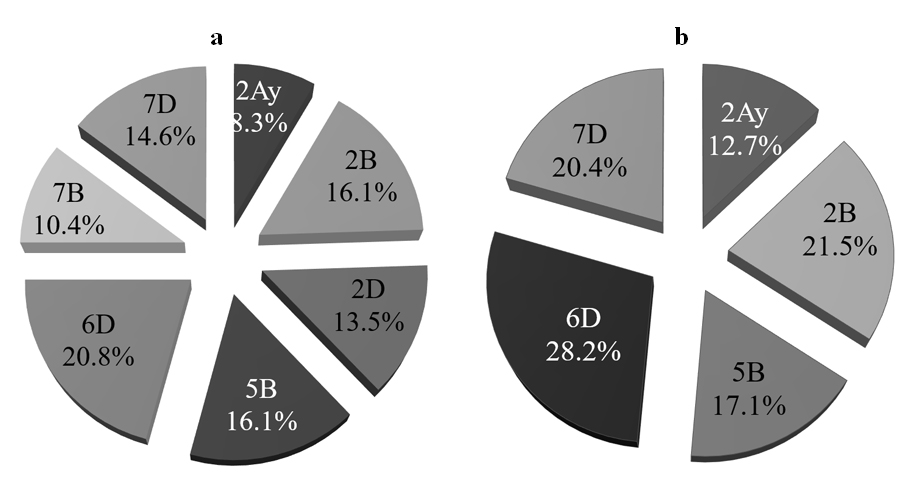 Figure 3. The frequency of retention of resistance loci (based on marker genotype). The analysis based on (a) the three populations “Yangmai#6” × “Mon/Ald”, “Yangmai#6” × “Chirya#3” and “Yangmai#6” × “Tia#1”, (b) the four populations (“Yangmai#6” × “Mon/Ald”, “Yangmai#6” × “Chirya 3”, “Yangmai#6” × “Tia#1” and “Yangmai#6” × “Ning#8201”.
Figure 3. The frequency of retention of resistance loci (based on marker genotype). The analysis based on (a) the three populations “Yangmai#6” × “Mon/Ald”, “Yangmai#6” × “Chirya#3” and “Yangmai#6” × “Tia#1”, (b) the four populations (“Yangmai#6” × “Mon/Ald”, “Yangmai#6” × “Chirya 3”, “Yangmai#6” × “Tia#1” and “Yangmai#6” × “Ning#8201”.
The frequency with which individual resistance genes were retained as a result of the selection procedure was tested in three of the four populations (“Yangmai#6” × “Mon/Ald”, “Yangmai#6” × “Chirya#3” and “Yangmai#6” × “Tia#1”). The “Ning#8201” chromosome 2D and 7B resistance genes were not included here, because their intrachromosomal locations have not yet been determined. The locus on chromosome 6D was present in 20.8% of the selections, while the chromosome 2B and 5B loci were each represented in 16.1% of the selections. The “Yangmai#6” chromosome 2A locus was inherited by 8.3% of the selections (Figure 3a). Among the “Yangmai#6” × “Ning#8201” selections, the most frequently retained locus was on chromosome 6D (28.2%), followed by that on chromosome 2B (21.5%) and that on chromosome 7D (20.4%). The locus on chromosome 2A was retained by just 12.7% of the selections (Figure 3b).
The improved level of spot blotch resistance expressed by the selections was likely the result of accumulating multiple genes. The lines, which have been developed here represent ideal donors of spot blotch disease resistance to Indian wheats, since they have simultaneously been selected for adaptation to the local environment. Of note is that their exploitation is facilitated by the availability of readily deployable SSR assays linked to the loci responsible for spot blotch disease resistance. Given that the parental materials were screened in successive years, it was possible to determine the extent to which the gain in resistance achieved by selection was an environmental rather than a genetic effect. Based on the performance of “Yangmai#6” over the two years, it could be concluded that the environmental component did not contribute significantly to the disease reaction. This conclusion was supported by the strength of the correlation between the disease reactions recorded for the F4 and F5 selections (data not shown).
The results showed that selection based on the SSR genotyping was successful in achieving genetic advance for resistance. In a population developed from the cross “Yangmai#6” × “Sonalika”, the chromosome 5B locus was found to be responsible for over 41% of the phenotypic variance for spot blotch resistance, while in a population developed from the cross “Ning#8201” × “Sonalika”, the chromosome 7D locus explained over 51% of the variance [18,21]. Combining both, the alleles in a single genotype not necessarily account for the phonotypic effect, equals to the sum of individual allele effect, i.e., 92% in this case. The QTL mapping results of Kumar et al. (2009) also showed that sum of individual allele is more than the combined effect [18].
Based on the indication that the number of genes are positively related to the strength of the resistance, the expectation is that there should be a benefit in stacking as many genes as possible if the aim is to maximize the level of resistance. Further, there are evidences that combining multiple genes not only ensure durable resistance across environments but also enhances resistance [30]. However, the accumulation of resistance genes beyond a certain limit appeared to no longer enhance the level of resistance as we also observed in some of the selections with comparatively lesser number of alleles but higher resistance. For example, selection #8 from the “Yangmai#6” × “Chirya#3” population carried four resistance genes/QTL (lying on chromosomes 2B, 2D, 5B and 7B), while selection #41 harbored five genes/QTL (chromosomes 2B, 2D, 6D, 7B and 7D); however, the level of resistance of these two selections was indistinguishable. Given that they have in common the loci mapping to chromosomes 2B, 2D and 7B, the inference is that there might be some QTL× QTL interactions where 6D is masking effect of 7D or vice-versa. There are several reports that describe QTL × QTLs interactions for polygenic traits including spot blotch [2,18,21,31].
Some of the selections exhibited a level of resistance similar to, or even inferior to that of their parents, despite harboring more resistance genes, while a few harbored a lesser number of resistance genes than did either of their parents, but nevertheless exhibited a higher level of resistance. A possible explanation for this apparent anomaly is the segregation of yet unmapped gene(s), which have remained undetected due to the inadequate genome coverage of current linkage maps and/or incomplete phenotypic characterization [32]. Spot blotch resistant selections in the genetic background of the successful Indian cultivar HUW234 have recently been developed by exploiting “Chirya#3” and “Ning#8201” as donors [33]. Both the Qsb.bhu-2A and Qsb.bhu-5B QTL have been incorporated using a marker strategy combining both foreground and background selection [33]. The approach adopted here differed in that the objective was to stack several genes to generate an effective source of near-immunity based on multiple genes.
Of the five resistance loci shared by all four of the populations, the chromosome 2B gene was retained more frequently (21.5%) than any of the others. This gene has been shown to be effective in a variety of genetic backgrounds [18,21]. The chromosome 6D locus contributes up to 22.5% of the variation for resistance [18], yet the selection regime appeared to favor it relatively heavily, implying some linkage to genes of importance to plant type. The chromosome 5B locus, although it has been classed as a major effect locus, was inherited by only one in six of the selections [18,34]. The current approach is slightly different from the Marker Assisted Selection (MAS), which is more effective for the minor effect QTL. Contrast to this, the phenotypic selection favors major effect QTLs/alleles [35]. Therefore, the 6D alleles might have been favored by the phenotypic selection in early generations since the selection in F3, F4 and F5 generations was partly based on agronomic appearance and spot blotch resistance. Overall, the use of a donor × donor crossing strategy was shown to be effective for developing lines with near-immunity to spot blotch disease, and by implication for enhancing the level of resistance to other diseases as well.
UK and AKJ designed the experiment, interpret the results and wrote the paper with the inputs from all authors. RP and SK made the crosse in the field. SK, RP and VKM conducted the field trials. MR and UK performed the molecular marker analysis. SK and UK analyzed the data. RC prepared the inoculum and evalauted lines in the field for spot blotch disease reaction.
The authors declare that they have no conflicts of interest.
Funds used for the acquisition of genotypic data were provided by the Government of India's Department of Biotechnology and the Indo-German BT/IN/Indo-German/10/UK2010 project. We thank Ram Awadh, Anette Heber and Sonja Allner for their technical assistance.
1.
2.
3.
4.
5.
6.
7.
8.
9.
10.
11.
12.
13.
14.
15.
16.
17.
18.
19.
20.
21.
22.
23.
24.
25.
26.
27.
28.
29.
30.
31.
32.
33.
34.
35.
Kumar U, Kumar S, Prasad R, Röder MS, Kumar S, Chand R, et al. Genetic Gain on Resistance to Spot Blotch of Wheat by Developing Lines with Near Immunity. Crop Breed Genet Genom. 2019;1:e190017. https://doi.org/10.20900/cbgg20190017

Copyright © 2020 Hapres Co., Ltd. Privacy Policy | Terms and Conditions